The main group elements tend to lose or gain the number of electrons needed to get the same number of electrons as the nearest noble gas.
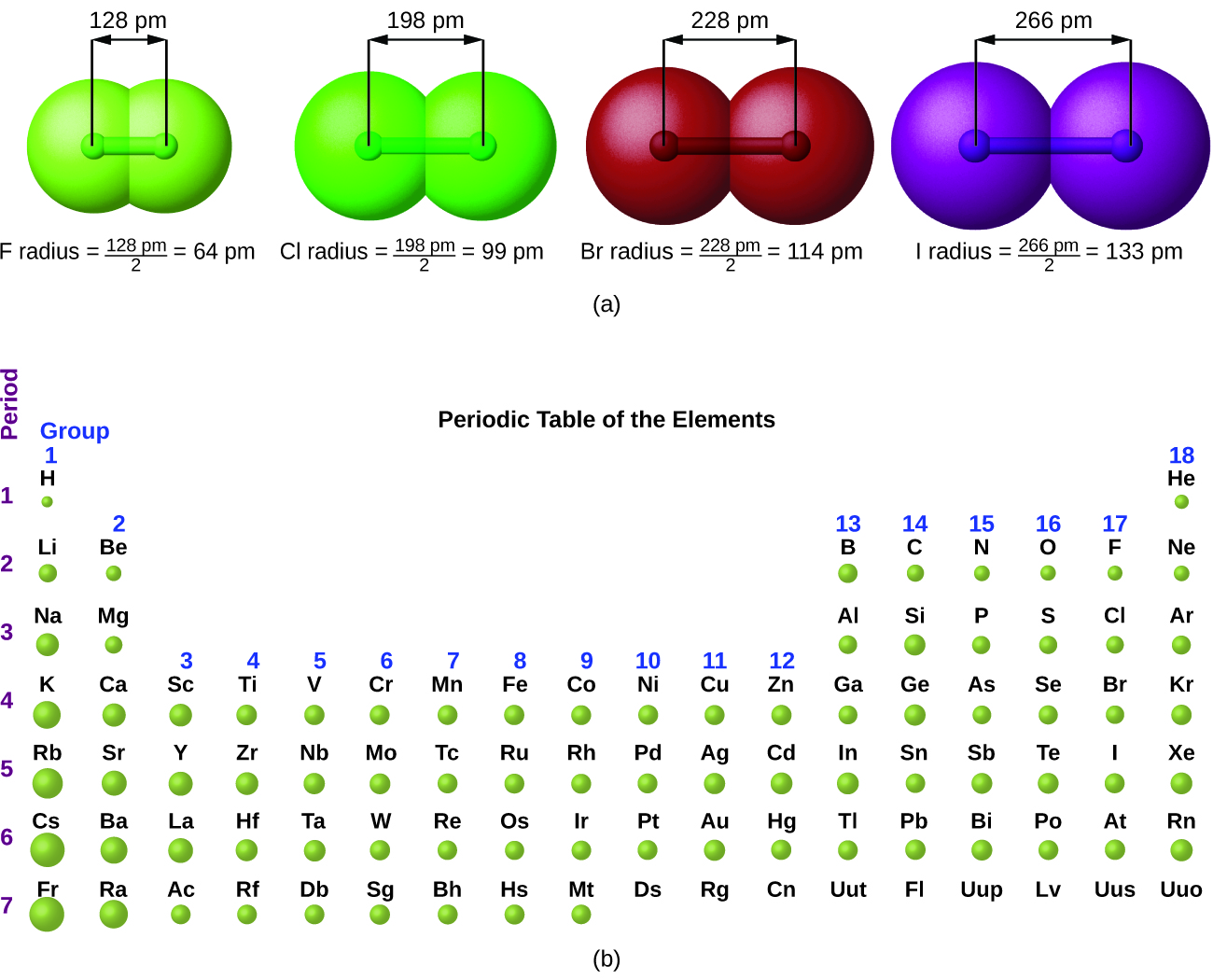
Z eff increases across a period ⇒ the atomic radius decreases across a period
The higher the effective nuclear charge Z eff, the more attracted the outer electrons are to the nucleus and the closer they are to the it.

The distance between the nucleus and the outermost electrons Elements of the same period have similar valence electron configurations and therefore behave in the same way during a chemical reaction The properties of elements depend on their valence electrons.

